 7-Methylxanthosine in turn is the precursor to theobromine, which in turn is the precursor to caffeine. Cleavage of the ribose and N-methylation yields 7-methylxanthosine. Theobromine is a purine alkaloid derived from xanthosine, a nucleoside. The mean theobromine concentrations in cocoa and carob products are: Item Theobromine can also be found in trace amounts in the kola nut, the guarana berry, yerba mate ( Ilex paraguariensis), Ilex vomitoria, Ilex guayusa, and the tea plant. Plant species and components with substantial amounts of theobromine are: Cocoa beans naturally contain approximately 1% theobromine. There are approximately 60 milligrams (1 grain) of theobromine in 28 grams (1 oz) of milk chocolate, while the same amount of dark chocolate contains about 200 milligrams (3 grains). There are usually higher concentrations in dark than in milk chocolate. Cocoa butter only contains trace amounts of theobromine. Theobromine is the primary alkaloid found in cocoa and chocolate. Chocolate is made from the cocoa bean, which is a natural source of theobromine. ĭespite its name, the compound contains no bromine, which is based on Greek bromos ("stench").Ī chocolate bar and molten chocolate. That name in turn is made up of the Greek roots theo (" god") and broma ("food"), meaning "food of the gods". Theobromine is derived from Theobroma, the name of the genus of the cacao tree, with the suffix -ine given to alkaloids and other basic nitrogen-containing compounds. Synthesis of theobromine from xanthine was first reported in 1882 by Hermann Emil Fischer. Theobromine was first discovered in 1841 in cacao beans by Russian chemist A. Related compounds include theophylline, caffeine, paraxanthine, and 7-methylxanthine, each of which differ in the number or placement of the methyl groups. 
It is also classified as a di methyl xanthine. Theobromine is a flat molecule, a derivative of purine. It is a white or colourless solid, but commercial samples can appear yellowish. It is found in chocolate, as well as in a number of other foods, including the leaves of the tea plant, and the kola nut. In industry, theobromine is used as an additive and precursor to some cosmetics. Theobromine is slightly water- soluble (330 mg/L) with a bitter taste. the signal of such an internal reference must not have its origin in two or more equivalent nuclei groups within the reference molecule.Theobromine, also known as xantheose, is the principal alkaloid of Theobroma cacao (cacao plant). The cause of this nonlinear behaviour is analyzed, and arguments are presented that it can be prevented by use of reference substances with only one single reference group per molecule, i.e. The failure of the AUS correction in producing parallel Scatchard plots from internally referenced shifts is caused by the nonlinear floating with the donor concentration of the internal reference signal. 115 l/mol), for the 4 apparent complex shifts (Icpt/K) and for the 4 nuclei specific AUS coefficients a 2 could be confirmed and somewhat improved by a Creswell-Allred processing of the data including AUS correction. 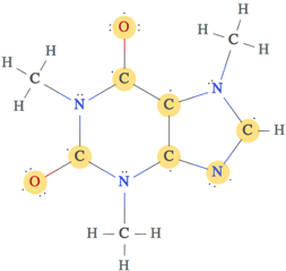
This gives evidence that the system behaves in accord with the pure 1:1 complex model and does not demand the assumption of simultaneous occurence of higher complexes. Equilibrium quotients K calculated from the externally referenced data for each of the 4 different protons were essentially the same. The plots for the 4 protons were parallel to one another in case of the externally referenced data and not parallel for the internally referenced data. Scatchard plots of the externally referenced data were curved upwards.Īpplying AUS corrections to both sets of data furnished linear Scatchard plots for ah signals. The 1-methyl plot even showed a maximum in the observed relative shifts ⊿ oi which turned to negative values for high benzene concentrations. Scatchard plots of the internally referenced data were curved downwards, the degree of curving increasing from 7-methyl/8-H to 3-methyl to 1-methyl. Shifts of the 4 signals were measured relative to both internal and external reference. The 4 proton signals of caffeine, dissolved in carbon tetrachloride, were investigated as functions of benzene concentrations in a very large concentration range. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
